Technology R&D
Botanical Drug Development
Introduction
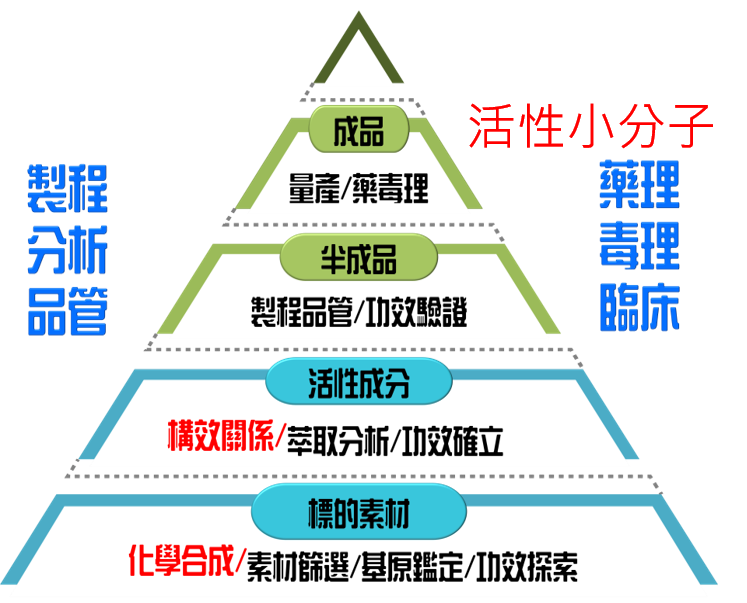
The research and development of herbal medicines prioritizes safety, efficacy, and controllable quality.
However, plants originate from nature and are influenced by environmental factors, making quality control extremely difficult. Therefore, the Pharmaceutical Technology Center's development of new herbal drugs involves not only research on plant raw materials, such as origin identification, chemical composition analysis, safety assessment, toxicity studies, academic literature, and even patent information, but also process exploration and optimization, coupled with pharmacodynamic and toxicological evaluations, to complete the development of new herbal drugs.
By establishing comprehensive preclinical CMC, pharmacological, and toxicological evaluation data, and further completing patent layout, not only is technological competitiveness effectively guaranteed, but traditional medicinal materials are also given scientific validation, laying the foundation for the internationalization of the herbal medicine industry.