
R&D
Technology R&D
Animal Toxicology
Introduction
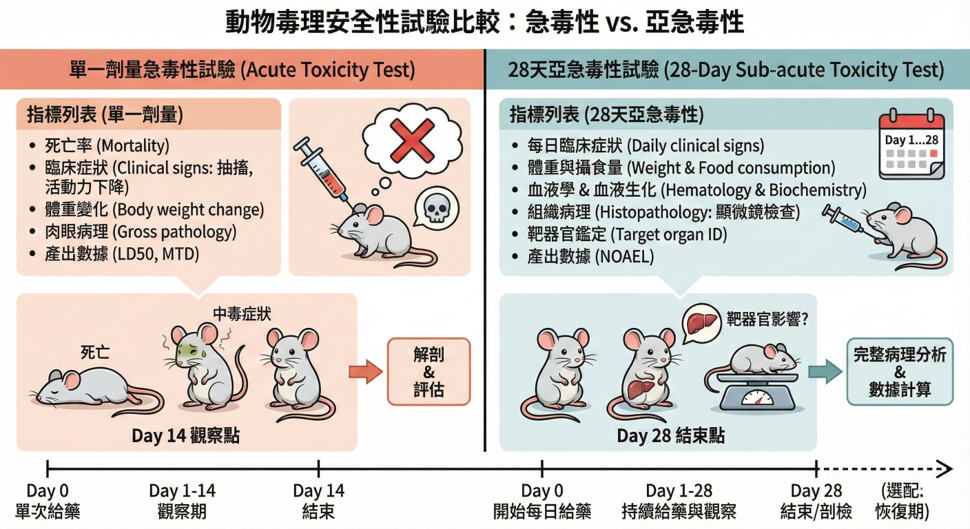
Acute and subacute toxicity studies in animals primarily assess the safety of the test substance to organisms under short- and medium-term exposure.
Acute toxicity studies observe animal survival, behavioral, and physiological changes after a single or short-term high-dose exposure; subacute toxicity studies administer multiple doses over a continuous period of 28 days or longer to assess organ toxicity, changes in blood and biochemical parameters, and to provide a basis for safe dosing and risk assessment.

Focusing on pharmaceuticals and medical devices, supplemented by health foods and medicated cosmetics, promoting technology development and services, dedicated to the development and internationalization of the pharmaceutical industry, enhancing industry competitiveness!
Contact Information
Copyright © 2026 Medical and Pharmaceutical Industry Technology and Development Center All Rights Reserved.